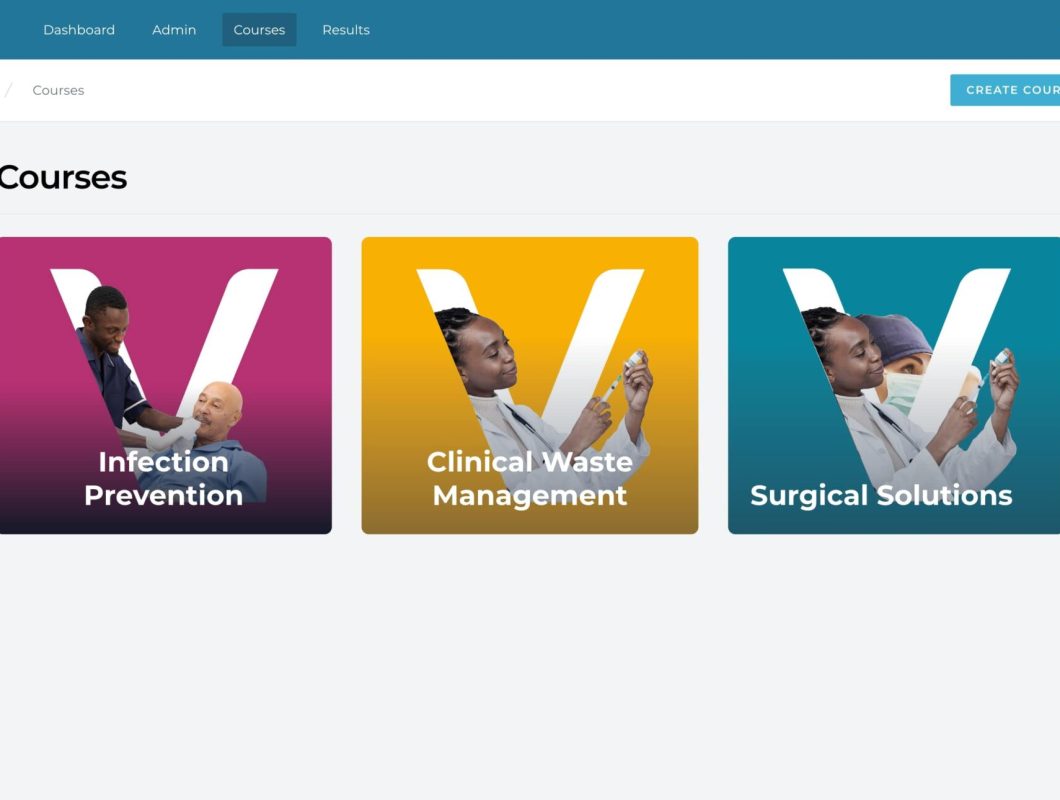
The first choice for single-use instruments.
Provide protection, precision, and performance every time with Instrapac®
The first choice for single-use instruments. Launched in 2002 with safety, convenience, and reliability in mind, the Instrapac® range offers the ideal solution for a wide range of clinical procedures, with both individual instruments and procedure packs available.
Why choose Instrapac® single-use?
Guaranteed sterility, every time
Our Instrapac® instruments and procedure packs are sterilised by us in-house, and offer you complete reassurance that the instrument you are using is sterile every time. Every patient, every procedure – always sterile.
Save time and improve efficiency
With no reprocessing or delays involved, our sterile instruments and packs support efficient, reliable and expert care in a range of procedures.1 Just open, perform, and dispose.Precision, Protection and Performance
Instrapac® instruments are designed to offer consistency and sharpness every time, with the confidence that every single instrument is brand new, sterile, and ready for use.

Trusted by
90% of the NHS
Instrapac® has been sold into 90% of the NHS in the past two years and is trusted by clinicians across the UK.
Saves valuable time
and improves efficiencies
Pre-packaged instruments and procedure packs, designed specifically for individual procedures, ensure you have all the tools you need in one handy, sterile pack, reducing set up and reprocessing time.1
Made from at least 80% recycled material
Our Instrapac® instruments are made from at least 80% recycled material, utilising products from other industries.
Improved
infection control
Our instruments and packs come sterile in an easy to open pouch, minimising the risk of cross-contamination and preventing infection.
Featured products

Instrapac® General Purpose Scissors
Product Code: 8572

Instrapac® Vaginal Examination Pack
Product Code: 7402

Instrapac® Delivery Pack Plus
Product Code: 8526

Instrapac® Podiatry Assistants Emery Pack – Soft Grip, Curved, Roller Spring, 14cm
Product Code: 8516

Instrapac® Nail Surgery Pack
Product Code: 7925
The majority of our Instrapac® range are Class IIa medical devices, and are fully certified by the British Standards Institution (BSI) to the regulation standards.

Contact us today about our Instrapac®
single-use instruments and procedure packs
Speak to our expert team to find out how we can support your instrument needs.
References
- NHS Supply Chain. (2024). Procedure Packs. Available online at: Procedure Packs framework agreement – NHS Supply Chain
News
Keep up to date with the latest Vernacare news
 Blogs
Blogs
Introducing the New Conti® Kind™ Collection
 News
News
Our Single Use Surgical Instruments website…
 News
News
Vernacare Acquires Eakin Surgical Limited
 News
News